Why Is Natco Pharma’s Stock Rising? Semaglutide Approval and Q3 Results in Focus
CDSCO nod for generic diabetes drug and 36% revenue growth drive over 9% intraday rally
Team Sahi
Natco stock moved sharply higher after regulatory approval for a diabetes drug and strong quarterly results. The surge followed confirmation of approval for a new diabetic drug and improved financial performance in Q3 FY26.
Shares of Natco Pharma rose more than 9% intraday. The movement reflected stock-specific developments rather than a broad sector rally.
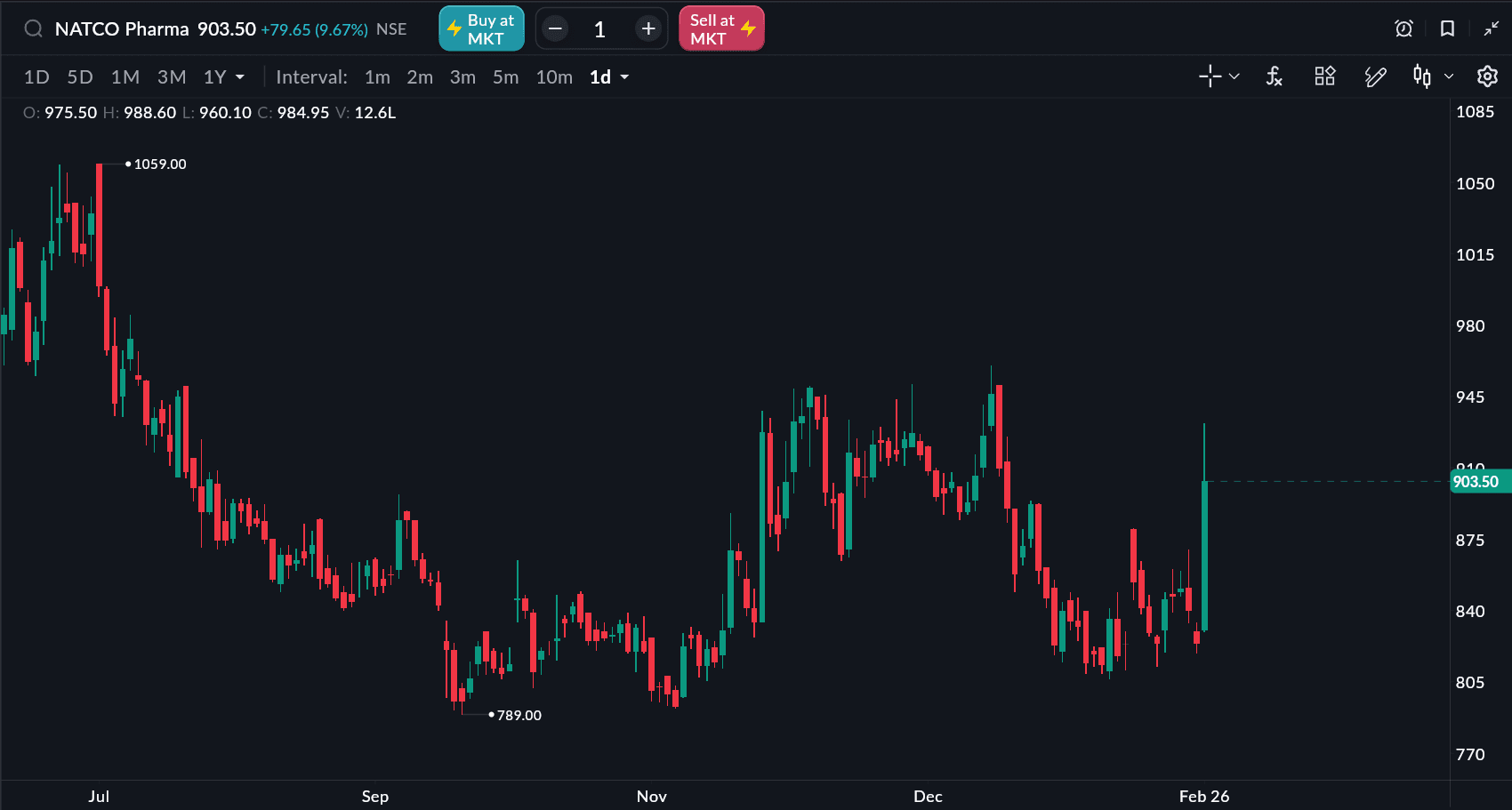
Natco Pharma Share Price Performance
As of 12:20 PM IST, Natco Pharma shares were trading over 9% higher, moving in the ₹919–₹928 range after a strong opening. The stock had previously closed at ₹823.85 in the last session.
Key price data:
-
Intraday gain: Around 9-10%
-
Intraday high: Above ₹932
-
52-week position: Around 13–14% below the 52-week high
The move placed the stock ahead of broader benchmark indices during the session.
Natco Drug Approval: CDSCO Clearance for Semaglutide
The key trigger behind the rally was a drug approval granted to Natco Pharma by the Central Drugs Standard Control Organisation (CDSCO) for a generic weight-loss drug used to treat obesity and metabolic disorders. Entry into the anti-obesity segment could offer a new revenue stream, especially as demand for GLP-1 based therapies continues to rise globally.
The approval, dated February 14, 2026, allows the company to:
-
Manufacture
-
Market
-
Launch generic Semaglutide injection in India
Semaglutide is used in the treatment of Type-2 diabetes. It belongs to a class of medicines known as GLP-1 receptor agonists.
What Is Semaglutide?
Semaglutide is prescribed to help control blood sugar levels in adults with Type-2 diabetes. It is commonly used along with diet and lifestyle management.
Globally, GLP-1 class drugs have seen rising demand. In India, diabetes prevalence has increased steadily, creating demand for more treatment options. The approval enables Natco Pharma to introduce a generic version in the domestic market.
The company has indicated plans for a March 2026 launch.
Financial Performance: Q3 FY26 Snapshot
Alongside the regulatory development, the company reported its Q3 FY26 results for the quarter ended December 2025.
Key Financial Metrics
|
Metric |
Q3 FY26 |
|
Net Profit (YoY) |
₹151 crore (+14%) |
|
Revenue (YoY) |
₹647 crore (+36%) |
|
EBITDA Margin |
24.5% |
|
Interim Dividend |
₹1.5 per share |
Revenue growth of 36% year-on-year indicates higher operational scale. Net profit growth of 14% reflects improved earnings compared to the same period last year.
The EBITDA margin of 24.5% shows operating efficiency during the quarter.
Impact on Generic Drug Stock Segment
Natco Pharma operates within the generic drug stock segment of the Indian pharmaceutical industry. Generic manufacturers often benefit from:
-
Regulatory approvals
-
Patent expiries
-
Domestic and export market access
Entry into a high-demand diabetes category expands the company’s domestic portfolio. The approval strengthens its product pipeline in a therapeutic area with sustained demand.
Additional Corporate Developments
During the quarter, the company also announced:
-
Leadership reappointment
-
Incorporation of a new subsidiary in Chile
The Chile subsidiary expands the company’s international footprint. Geographic diversification can support export-oriented operations in regulated markets.
Why Regulatory Approvals Affect Pharma Stocks
In the pharmaceutical sector, regulatory decisions can materially influence stock movement. Approvals determine whether a company can commercialise a product.
Key effects of drug approvals:
-
Expansion of product portfolio
-
Access to new revenue streams
-
Entry into new therapeutic categories
-
Strengthening of domestic or export presence
In this case, the Natco drug approval for a new diabetic drug enabled entry into the GLP-1 category in India.
Broader Context: Diabetes Market in India
India has one of the largest populations of people with diabetes globally. Demand for anti-diabetic medication has grown over time.
GLP-1 receptor agonists are increasingly used in diabetes treatment. Generic availability can influence pricing and accessibility within the domestic market.
The planned March 2026 launch positions the company within this growing therapeutic segment.